The students will share thier presentations with the class and post them to the class web site. They will utilize the software to create a presentation including the information stated in the rubric for each element.
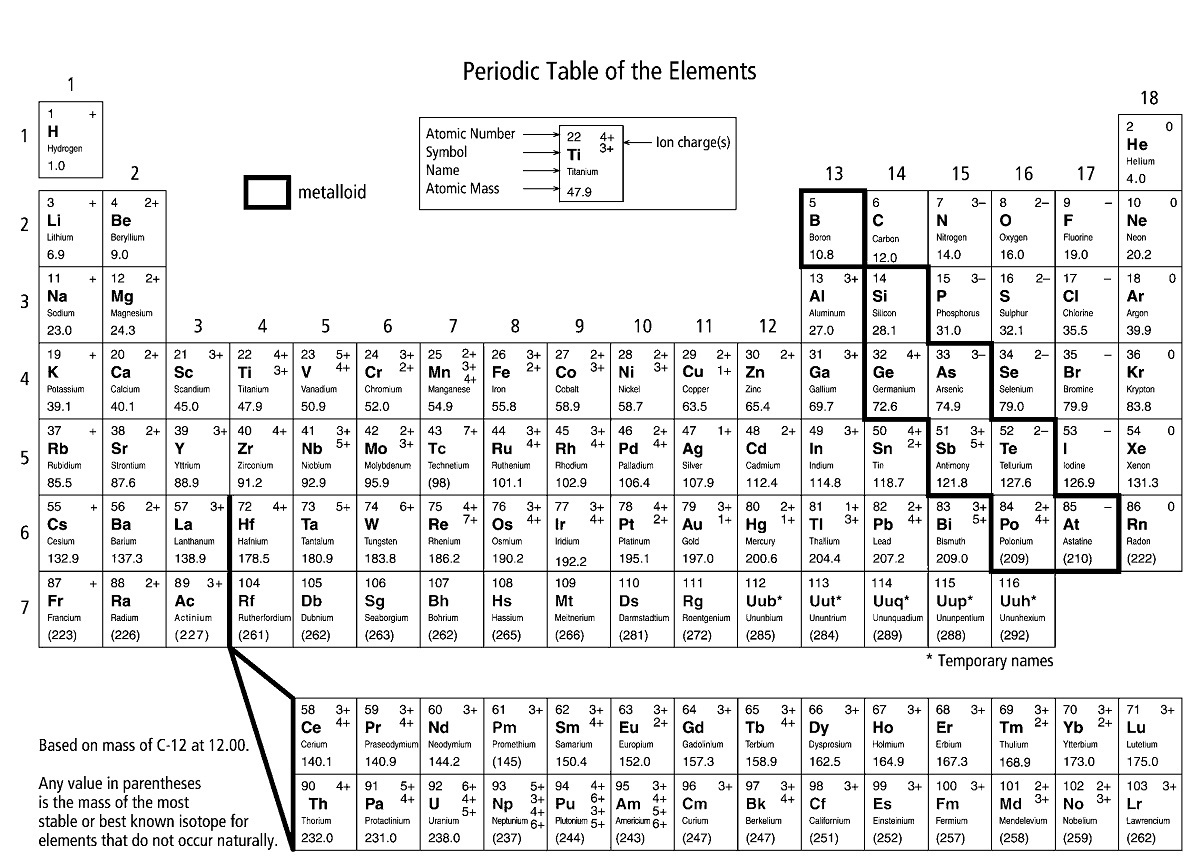
The students should include which atoms are grouped together by valence electrons. The number of electrons in the outer shell (valence electrons) and the Lewis dot structure will also be included. Students will create a power point presentation of the first 20 elements including the name, symbol, number of protons, neutrons and electrons.

Students will access the build an atom site at to review the structure of atoms and how they are organized by protons and outer electrons.Students will view the following you tube video to understand the periodic table of elements:.The teacher will demonstrate the organization of the periodic table using the wall poster.Ĭomputers with basic office applications installed as well as internet access.
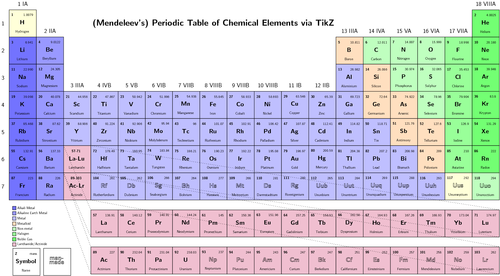

Objective 3: Students should be able to identify the electron configuration for a given elements based on its location on the periodic table.Ī large periodic table is displayed in the front of the classroom. Objective 2: Students should be able to categorize elements based the relationship between periods and energy levels for the first 20 elements. Objective 1: Given a diagram of the periodic table, the students will be able to identify and differentiate between groups and periods.
